Edwards Lifesciences at PCR London Valves 2024
Don't miss Edwards Lifesciences at PCR London Valves, featuring the latest transcatheter devices for high quality treatment of heart valve disease1,2
Edwards Scientific Program
Join us if you want to:
- Learn how latest generation of transcatheter heart valves can facilitate a contemporary lifetime management of patients with valvular heart diseases
- Discuss the latest clinical outcomes data showing how transcatheter heart valves technologies can enhance the quality of patient care
- Appreciate how a portfolio of device options can be employed to treat a broad spectrum of valve anatomy
Agenda
| Session objectives Sam Dawkins |
| A portfolio of options to treat patients with mitral and tricuspid regurgitation Volker Rudolph, Philipp Bartko |
| Discussion and audience interaction All |
| Transition to investigational device Nicolas Van Mieghem |
| Recorded case: Transseptal mitral valve replacement John Webb |
| Closing and transition to TAVI session Radoslaw Parma, Nicolas Van Mieghem |
| The future of lifetime management Francesco Saia |
| Discussion and audience interaction All |
| Patient presentation Radoslaw Parma |
| LIVE Case procedure Tiffany Patterson, Simon Redwood |
| Session evaluation and key learnings Sam Dawkins |
Speakers

Prof. Sam Dawkins
John Radcliffe Hospital, Oxford, UK

Prof. N.V. Mieghem
Erasmus University Medical Centre, Rotterdam

Dr. Radoslaw Parma
Medical University of Silesia

Prof. Volker Rudolph
Bad Oeynhausen, HDZ

Priv.-Doz Philipp Bartko
Medical University of Vienna

Prof. Francesco Saia
University Hospital of Bologna

Prof. Giuseppe Tarantini
University Hospital of Padua

Prof. John Webb
St Paul's Hospital Vancouver

Dr. Tiffany Patterson
St Thomas' Hospital London

Prof. Simon Redwood
St Thomas' Hospital London
Join us if you want to:
- Discover the results of the latest randomised clinical trials in TAVI: EARLY TAVR and RHEIA
- Learn how these new data may impact clinical practice guidelines
- Discuss what will be the new frontiers in TAVI including the treatment of patients with bicuspid aortic valve and moderate aortic stenosis
Agenda
| Welcome and session objectives Victoria Delgado |
| EARLY TAVR: what you need to know Philippe Genereux |
| How will the EARLY TAVR findings impact on clinical guidelines? Hendrik Treede |
| New frontiers in asymptomatic patients: case examples Julia Mascherbauer |
| Discussion and audience interaction All |
| RHEIA: what you need to know Helene Eltchaninoff |
| How will the RHEIA findings impact on clinical guidelines? Hendrik Treede |
| New frontiers in female patients: case examples Mariuca Nicotera |
| Discussion and audience interaction All |
| What are the unmet clinical needs in TAVI? Philippe Pibarot |
| Discussion and audience interaction All |
| Session evaluation and key learnings Victoria Delgado / Bernard Prendergast |
Speakers

Prof. Victoria Delgado
Germans Trias i Pujol Hospital, Barcelona

Dr. Philippe Genereux
Morristown Medical Center, NJ

Prof. Bernard Prendergast
St. Thomas' Hospital - London

Prof. Helene Eltchaninoff
University Hospital of Rouen

Prof. Hendrik Treede
University Hospital Halle - Mainz

Prof. Mariuca Nicotera
Hospital Sindelfingen

Prof. Julia Masherbauer
University Hospital St. Pölten

Prof. Philippe Pibarot
Quebec Heart and Lung Institute
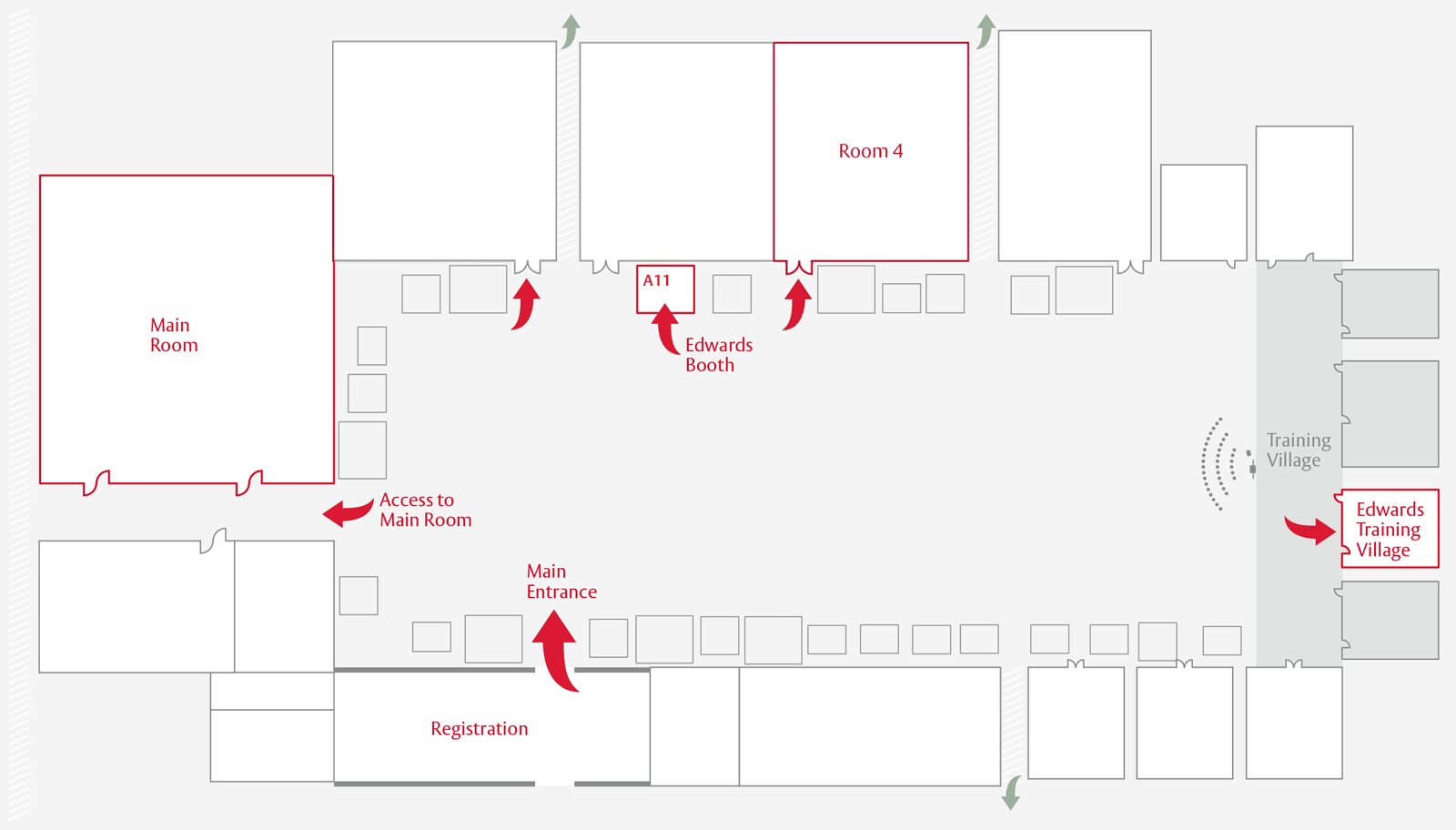
Finding us at PCR London Valves 2024
Lifetime Management Masters – Challenging cases performed with SAPIEN 3 Ultra RESILIA valve and SAPIEN 3 Ultra valve
Join us for two unique case presentations at the Edwards booth. You will discover how young promising implanters successfully managed challenging TAVI cases with the SAPIEN 3 Ultra and SAPIEN 3 Ultra RESILIA valves.
Dates:
Sunday, November 24th - 15:45–16:15
Monday, November 25th - 10:30–11:15
Edwards Training Village
Join us at the training village to learn more about the latest TAVI implantation with the SAPIEN 3 Ultra RESILIA valve on aortic and mitral simulators. We will enable you to practice your technique on different configurations.
Register for News and Post Event Content
Sign up now to receive copies of Edwards Lifesciences recorded event sessions post event, as well as receive event and news information from Edwards Lifesciences, including:
- Educational videos and articles
- Publications
- Congress highlights
- Webinar invites
- Patient support tools
- Product announcements and updates
heading 3
Content goes here
No clinical data are available to evaluate the long-term impact of RESILIA tissue in patients. Additional clinical data for up to 10 years of follow-up are being collected to monitor the long-term safety and performance of RESILIA tissue.
References
- Mack MJ, et al. PARTNER 3 Investigators. Transcatheter Aortic-Valve Replacement in Low-Risk Patients at Five Years. N Engl J Med. 2023 Nov 23;389(21):1949-1960.
- Stinis CT, Abbas AE, Teirstein P, et al. Real-World Outcomes for the Fifth-Generation Balloon Expandable Transcatheter Heart Valve in the United States. JACC Cardiovasc Interv. 2024.
Medical device for professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
